As one of the fastest growing segments of agricultural inputs, biological products are typically categorized as biostimulants, biopesticides or biofertilizers. The active ingredients in biologic products, by definition, are derived from naturally occurring microorganisms, plant extracts, beneficial insects or other organic matter.
The nature of living organisms and natural chemistries in biologic products present unique challenges in the product evaluation and field development process as well as regulatory hurdles. Recognizing and addressing these challenges with a plan to deliver a strong product position backed by consistent and reliable trial data can produce valuable results. A well-planned approach like the AgriThority Prescriptive Response™ development service also helps avoid the pitfalls that could delay the product development process and market entry as well as increase costs.
Challenges often arise when initiating a trial in a different country, either for counter-season data generation or local product development. Expertise in protocol development can reduce the number of total trials and seasons required to evaluate performance and gather data for product registration. Proficient analysis can increase the value of the information. Inefficiencies result in less reliable data and may require supplemental trials to support the project goal.
AgriThority® has tested biostimulants and biofertilizers in more than 800 protocols around the world over the past ten years, with proven experience meeting demands in regulatory and product development processes across many geographies. Through implementation of trial designs and protocols specific to this category of products we can more accurately evaluate performance and accelerate the product’s path to commercialization. Use of innovative research tools also aid in the development of Best Management Practices to guide expectations and successful use by the growers.
Early Trial Planning
Early project planning can have a significant impact on the effectiveness of a trial. An adequate efficacy evaluation involves an optimal timing window to allow full expression of product effects on the target crop. But, the first steps are to start early and to ensure regulatory compliance.
A biologic or biostimulant product imported to a field location from another country requires regulatory permits. South America may be used as an example. While these products may gain importation approval to, for example, Paraguay in less than one month, Brazil and other countries may take six months or more to process. Ignoring this additional time in planning can imply a field trial start under non-relevant environmental conditions, compromising future use of the data generated in the project. In many countries, new biostimulant or biofertilizer products are not adequately covered in existing local regulatory legislations, thus permit and registration processes are not completely clear. In these situations, regulatory experience and fluent communication with the importing country’s government institutions can streamline the application process and expedite permit approval.
When testing biologic products, consider storage and handling requirements of products containing living organisms during shipment and at the testing location. In some countries, customs release can involve a significant period. If the product requires special conditions, be sure these can be followed during the shipping process and by the trial organizer. Requirements may include refrigeration of the product prior to use, or application as a seed treatment shortly before planting. For biologicals, including a viability test in the protocol may be a good check to ensure proper product quality in each experiment.
Early Development Trials
New technology testing often jumps “from lab to field,” skipping the early development trial which allows for a better understanding of its effects on crops. Early development trials fine-tune aspects of future protocols by measuring early crop growth. Conducted in a greenhouse or as a closely controlled plot trial, the early development study is relatively low cost and short duration yet increases the effectiveness of future field trials significantly. It provides a better understanding of the product by discovering the optimal application rate as well as compatibility and synergies with other technologies normally used in the target crops.
Early development trials can be an effective source to substantiate product claims and complement the data needed in registration application. The early development stage provides an opportunity to show how the biologic product performs in coordination with other biologic or synthetic chemistries through detailed measurements. Testing includes controlled levels of biotic and abiotic stress factors that can provide an initial understanding of the scope of product effects. This is often difficult to perform in larger scale field trials at early stages.
Location Selection
Trial scope is typically limited by cost constraints; therefore, each trial should be designed to produce a maximum amount of information for product development and registration. The optimal design of a field trial is strongly influenced by the environments which best demonstrate product performance or match conditions experienced by the target grower (e.g., a stronger relative product effect under low-yielding environment).
A careful choice of trialing environments may consider the soil type, temperature range, probability of drought and heat stress, plus pest and disease infestations and other factors that may affect expression of product effect on the crop. Trial organizers may have capabilities to further control stress factors through irrigation systems or retractable roof structures under well calibrated protocols.
Attempting to measure product response to too many biotic and abiotic environmental factors in a single project is normally tempting but may create inefficiencies when considering the typical range of responses. When focusing on stress environments, trialing efforts should account for a higher relative experimental error. Thus, it is wise to adequately plan the total required number of locations.
Tools to Measure Plant Performance
Data generated by the trial is amplified through advanced tools used to quantify crop response. Measurable data permits objective product comparisons, more interpretable and credible by growers than subjective information sources dependent on the “eye test.”
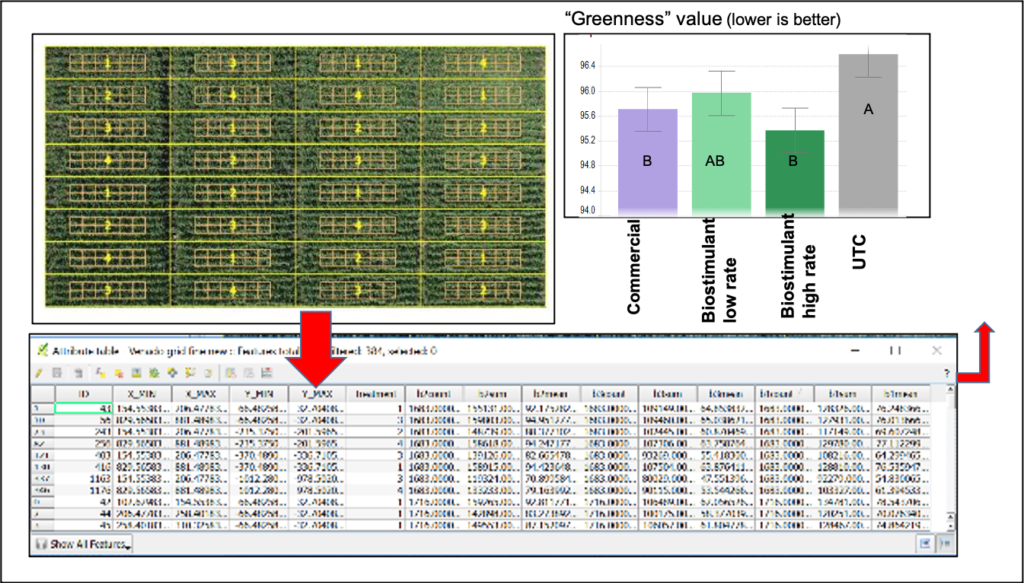
Examples of advanced trial evaluation tools are ground or aerial (drone, plane or satellite)-based Normalized Difference Vegetation Index (NDVI) sensors, which provide a visual snapshot of the greenness or indices closely related to crop health and photosynthetic activity in the trial at a given point in time. When overlaid with the experiment layout grid (Figure 1), a software process converts the aerial image to measurable values to compare products within the trial.
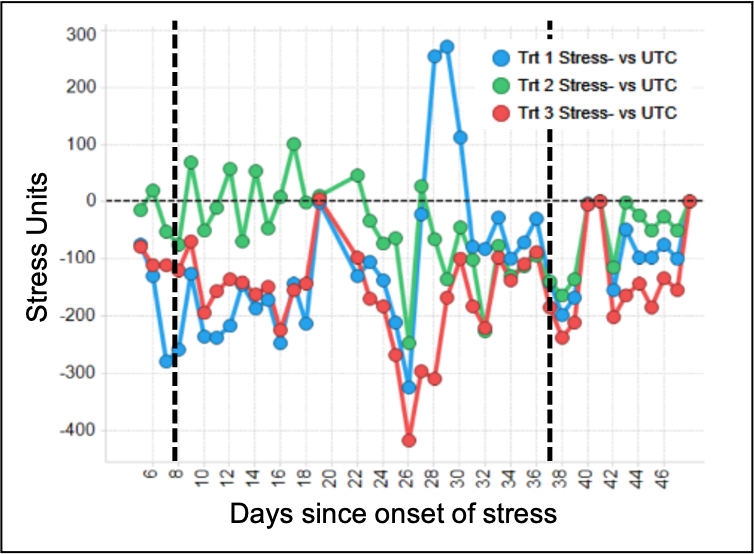
Similarly, a quantitative estimation of plant size and leaf area at early growth stages can be achieved through use of simple digital image processing software. A remarkable example in our evaluation of responses and characterization of biostimulants, biofertilizers and biopesticides has been the use of sensors for crop canopy temperature estimation under drought or nutritional stress. The sensors are positioned in the field to collect multiple data points each day along the season to detect possible response from treatments that improve crop tolerance to stress (Figure 2). As the crop water status and plant health improves, the sensors record a lower leaf temperature. Measurement of root structure can be achieved through samples removed from the field during the trial and further processing through digital images and conversion to numerical data through software, as well.
Analysis is Key
Once the trial is complete, statistical analysis and adequate use of data visualization tools convert data into usable information and actionable next steps. A careful inspection of the dataset and a robust analysis ensures lack of confounding effects that may hinder or bias estimation of product performance. The product development team relies on the analysis to determine whether to continue developing the product or proceed to registration. Once the biofertilizer or biostimulant is available to growers, data can substantiate claims and validate product performance. After commercialization, continuous trialing can show product consistency over several seasons, performance in new environments, or integration with newer technologies.
Well-designed trials performed on schedule with qualified suppliers can increase efficiencies and control costs in product development. Through early planning and efficient implementation of trials, companies often can accelerate the introduction of new biostimulant and biofertilizer technologies to market and encourage use of more sustainable agricultural production practices.
For more information, contact us through AgriThority.com or call +1-816-891-0916



